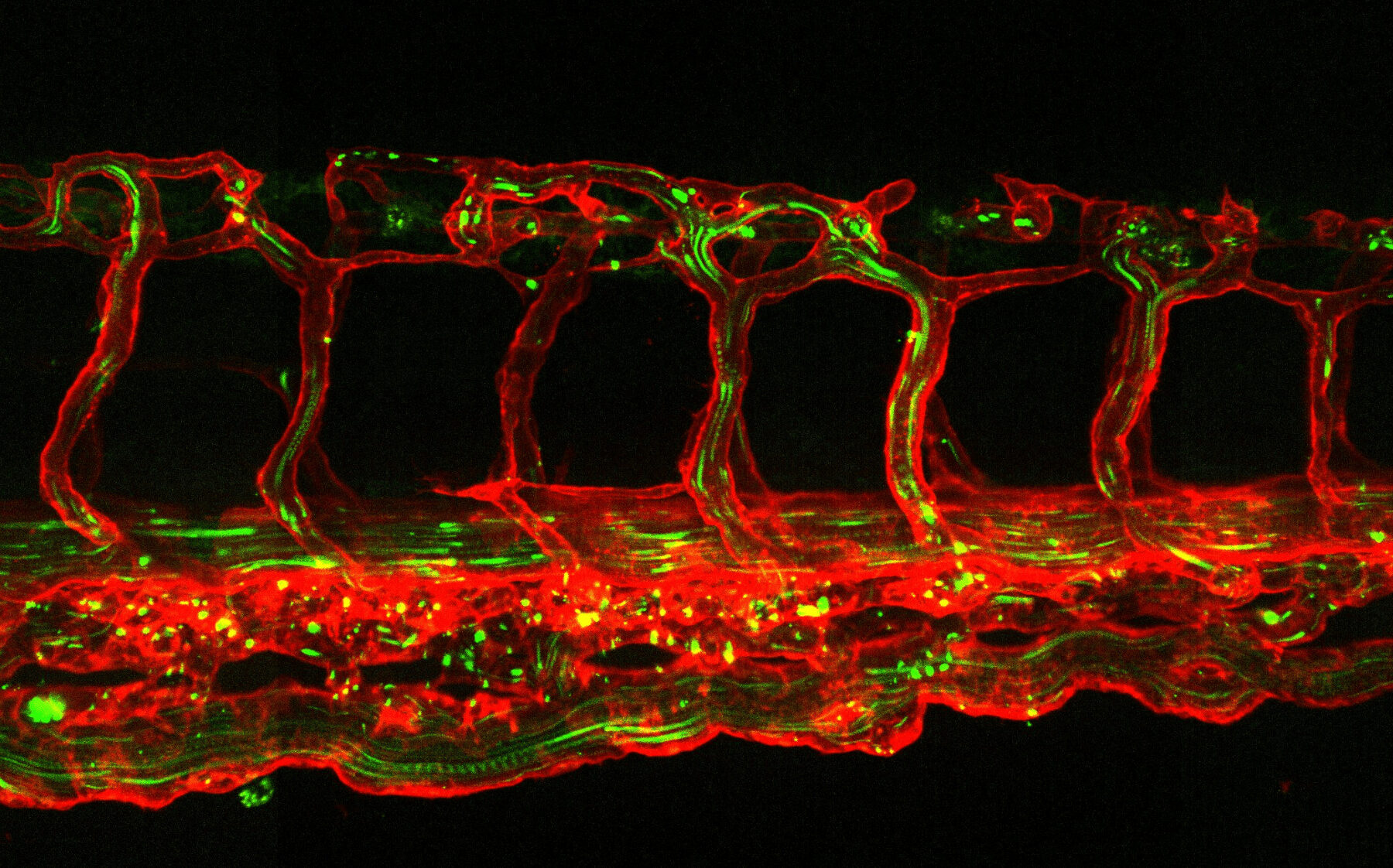
The Alsace node regroups 6 Life Sciences imaging facilities in Strasbourg, Illkirch and Mulhouse and six highly visible R&D teams expert in microscopy techniques and image analysis tools. The Alsace node offers a high level of technical and innovative methodological expertises in multi-scale imaging at the interface between biology, chemistry, optics and physics. The node aims to offer a fully integrated biological imaging portfolio from the molecule to the small animal/plant within the FBI infrastructure, based on its innovative chemical, optical and biocomputational developments, as well as on its already established network of state-of-the-art imaging instruments (super-resolution and quantitative fluorescence microscopies, confocal microscopies, F-techniques, small animal imaging systems (SPIM, macroscopes, OCT…)).
2022 in Numbers
- 336 hosted projects
- 90 publications
- 15 training programs
- 17 patents since 2011
Technological Innovations
- Tomographic diffractive microscopy (TDM) has been developed for 3D label-free imaging at cellular level, with an improved resolution compared to conventional microscopes, and, not being limited by possibly weak fluorescence, potentially at high speed (several 3D images/s).
- Single particle tracking and time-resolved luminescence microscopies have been developed to image upconverting nanoparticles (UCNPs). Due to their anti-Stokes emission, UCNPs allow imaging applications with exceptional signal to noise ratio.
- Single-shot full-field optical coherence tomography combines FF-OCT with off-axis interferometry. It extracts an FF-OCT image from a single interference acquisition, enabling single-shot high-resolution imaging within turbid media such as biomedical samples.
- Development of an integrated software for image reconstruction, correction, co-localization, resolution estimation, segmentation and clustering of labelled complexes (SharpViSu & ClusterViSu), including 3D analysis and segmentation of SMLM data using Voronoi diagrams.
- Molecular and supramolecular complexes have been developed for anti-Stokes imaging at the molecular level.
Tech transfer
- Single particle tracking and time-resolved luminescence microscopy of UCNPs
- Ultrabright lanthanide-based nanoparticles for diagnostic
- Plasma membrane probes (Membright) & Polarity-sensitive membrane probes (NR12S, NR12A, NR4A)
- Transmission Tomographic diffractive microscopy
- Integrated software for image reconstruction, correction, co-localization, resolution estimation, segmentation and clustering of labelled complexes (SharpViSu & ClusterViSu)
Most Innovative Systems Available for Booking
- Lightsheet microscope
- Super-Resolution (SMLM, STED)
- Electron Microscopy (Serial Block Face, Tomography, Cryo)
- Molecular Imaging (FCS, FLIM, anti-stokes)
- Live Imaging (Spinning disk)
QuESt (Quantum Efficiency Strasbourg)

Facility: QuESt (Quantum Efficiency Strasbourg)
Head: Ludovic Richert & Bertrand Vernay
The QuESt imaging facility combines the microscopy resources of the Institut de Génétique et Biologie Moléculaire (IGBMC) and the Laboratoire de Bioimagerie et Pathologies (PIQ). The two laboratories are located on the Illkirch bio-campus, just 10 minutes’ walk from each other. QuESt has held the IBiSA label since 2014. QuEst offers a range of instruments for multi-scale imaging, from the molecule to the whole animal. The ICI (Imaging Center IGBMC) component located at the IGBMC specialises in imaging the dynamic processes of living organisms at the molecular, cellular and whole organism levels. Researchers can analyse, in an integrated manner, their study models at different resolutions, ranging from the finest cellular structures to the complex functioning of organs in vivo. The PIQ (Plateforme d’Imagerie Quantitative – Quantitative Imaging Platform), located in the Faculty of Pharmacy, has a specific focus on quantitative molecular microscopy methods. In addition to commercial instruments, the PIQ-QuESt develops its own state-of-the-art instruments.
Microscopy systems available @QuESt
Services on this Facility
Plateforme d’Imagerie Cellulaire de l’IBMP

Facility: Plateforme d’Imagerie Cellulaire de l’IBMP
Head: Jerome Mutterer & Mathieu Erhardt
The microscopy facility was the first technology platform established in the IBMP in 1998. Its scientific programme aims at understanding the expression of plant or animal genes over space and time at various levels. Microorganisms or biomaterials studied by partner research units are other topics of interest. Our facility follows official guidelines for « Plates-Formes Technologiques du Vivant » and has received RIO 2001, 2004 and 2006 labels. It’s part of the larger PIC.sc Strasbourg Centre Cell Imaging Facility that allows sharing devices and knowledge from several research units from CNRS, INSERM, UNISTRA. Our missions include assisting research from IBMP and partner research units, developing and implement new imaging technologies, training our user base and beyond, getting involved in microscopy education and science popularization.
Microscopy systems available @Plateforme d’Imagerie Cellulaire de l’IBMP
Plateforme Imagerie In Vitro @INCI

Facility: Plateforme Imagerie In Vitro @INCI
Head: Sylvette Chasserot-Golaz (LM) & Frank Pfrieger (EM)
The platform is attached to ITI Neurostra. It is located at the Institut des Neurosciences Cellulaires et Intégrées (INCI) and managed administratively by the CNRS UAR 3156 headed by P. Darbon.
The expertise of the in vitro imaging platform applies to the study of biological materials, tissues and isolated cells on a structural and ultrastructural scale. It extends to the immunocytochemical visualisation of molecules and the phenotypic detection of gene expression.
The platform is involved in a wide variety of interdisciplinary projects (biology, chemistry, agronomy, medicine). Around two-thirds of users come from the CNRS INCI UPR3212 (6 groups), with the remainder coming from other Strasbourg institutes (IBMC, IBMP, ISIS, IPHC, ECPM, CRBS, etc.).
A number of developments have taken place in recent years, including cryofixation (Wohlwend system) and super-resolution microscopy (Stellaris 8 confocal microscope with a STED module).
Microscopy systems available @Plateforme Imagerie In Vitro INCI
PI2 (Plateforme d’imagerie de l’I2CT)

Facility: PI2 (Plateforme d’imagerie de l’I2CT)
Head: Jean-Daniel Fauny
The PI2 imaging technical platform belongs to the UPR3572 research unit: I2CT (Immunology, Immunopathology and Therapeutic Chemistry) of the IBMC (Institute of Molecular and Cellular Biology) in Strasbourg. The platform has i) sample preparation tools, ii) microscopy/imaging tools and iii) image processing and analysis tools. The platform offers workflows involving these 3 stages in order to answer the biological questions raised by scientists. The platform is part of the local RISEst network (Réseau d’Imagerie Strasbourg grand Est) and works in close collaboration with the other platforms in the network.
Microscopy systems available @PI2
PIC-STRA (Plateforme d’Imagerie du CRBS – STRAsbourg)

Facility: PIC-STRA (Plateforme d’Imagerie du CRBS – STRAsbourg)
Head: Pascal Kessler
PIC-STRA aims to support the 10 CRBS research units, as well as external teams from both the academic and private sectors. Opened in October 2020, this 350 m2 imaging platform provides users with around ten imaging systems (stereomicroscopes, wide-field and confocal single- and multiphoton microscopes with super-resolution module) for multi-scale observation, from whole small animals to sub-cellular details. It provides various solutions for the observation of fixed and living samples (videomicroscopy) and is equipped for image processing and analysis (IMARIS, Fiji/ImageJ, ICY, iLastik). The platform is part of the local RISEst network (Réseau d’Imagerie Strasbourg grand Est), is in the process of obtaining CORTECS and IBISA (STrasbourg Centre) accreditation and works closely with the other platforms in the network.
Microscopy systems available @PIC-STRA
Plateforme de Microscopie confocale @IS2M

Facility: Plateforme de Microscopie confocale @IS2M
Head: Tatiana Petithory
The ISO 9001 certified platform consists of 2 systems:
- Zeiss Axio Imager M2 LSM 800 confocal upright microscope with PI XY motorized piezo stage, 2 PMTs and an AiryScan super resolution module The excitation wavelengths available on the equipment are 405nm, 488nm, 561 nm, 640 nm. An OKOLAB incubation chamber maintains a stable temperature and CO2 level in the microscope for live image acquisition. This equipment allows the acquisition of topography information by reflection and fluorescence simultaneously.
- Inverted confocal microscope Spinning disk CSU W1: The stand is an Eclipse Ti2-E Nikon equipped with a motorized XY stage and a piezzo Z-stage, the “Perfect Focus” focus maintenance system and a “water dispenser immersion”. The confocal spinning disk is the Yokogawa mono-camera SD CSU-W1 mono-disk (50µm), the Oxxus excitation laser source has 4 wavelengths 405-488-561-640 nm. The sCMOS Hamamatsu Orca Flash 4.0 camera complements this detection equipment as well as a thermostated and CO2 OKOLAB incubation chamber with different insert sizes. An off-line station allows the analysis of images with the software ZEN blue, NIS-AR, FiJi, ICY and Imaris and to manipulate large files. A reservation system has been also set up.
Microscopy systems available @IS2M
Microscopic Imaging & Image Treatment team @IRIMAS

R&D team: Microscopic Imaging & Image Treatment team @IRIMAS
Head: Olivier Haeberlé
IMTI team develops innovative microscopes to image unlabeled samples. Such systems achieve 3D refractive index distribution measurements at cellular level, with twice-improved resolution compared to conventional optical microscopes. We have achieved several premieres in the domain:
2009: world-first TDM with improved resolution: 130 nm, world record at that time (Opt. Lett. 34, p. 79 (2009)).
2010: world-first images in correlative TDM-Fluorescence microscopy (J. Biophoton. 3, p. 462 (2010)).
2010: world-first dual transmission-reflection TDM images (J. Mod. Opt. 57, p. 740 (2010)).
2014: world-first high lateral resolution/high longitudinal precision profilometry (Appl. Opt. 53, p. 748 (2014)).
2017: world-first improved and isotropic resolution TDM images (≈180 nm in 3D, distinguishing absorption and refraction. This achievement was awarded the cover of Optica, April 2017 (Optica 4, p. 460 (2017)).
2019: world-first experimental mirror-assisted TDM images (albeit in simplified configuration) (J. Opt. Soc. Am. A 36, COF1 (2019)).
Biophotonics of molecular and cellular interactions team @LBP

R&D team: Biophotonics of molecular and cellular interactions team @LBP
Head: Yves Mély
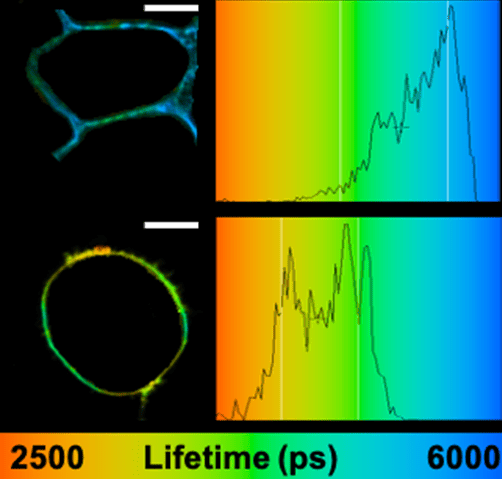
The highly multidisciplinary Biophotonics team develops fluorescence-based imaging techniques and innovative probes, in order to apply them for solving key questions related to the molecular and cellular functions and interactions of viral and epigenetic proteins with their nucleic acid targets. The physicists and engineers of the team have implemented and developed home-made microscopy set-ups, which are regularly upgraded and optimized for new applications and projects. The team implemented in 2003 one of the first two-photon FLIM/FCS microscopes in France. Later, the team developed a high resolution 3D dSTORM/PALM microscope and a STED microscope with cw laser. In order to track and image UCNPs, an anti-Stokes wide-field for SPT and an anti-Stokes luminescence lifetime confocal microscope were developed. More recently, a spectral PAINT (sPAINT), a light sheet single molecule microscope and a single molecule UV microscope were developed. These instruments are accessible to the community through their integration into the platform PIQ-QuESt (IBISA).
Nanochemistry and Bioimaging team @LBP

R&D team: Nanochemistry and Bioimaging team @LBP
Head: Andrey Klymchenko
The team develops multifunctional fluorescent molecules and nanoparticles for biomolecular detection and imaging. We developed i) solvatochromic probes for ratiometric and super-resolution imaging of plasma membrane organization and cell polarity mapping, and ii) fluorogenic membrane probes operating from green to near-infrared range for imaging cells, tissues and organoids by advanced microscopy techniques. We also introduced environment-sensitive fluorogenic probes for imaging G protein coupled receptors and intracellular RNA. We also developed dye-loaded fluorescent polymeric nanoparticles (NPs) 100-fold brighter than quantum dots of similar size. They were successfully applied for single-particle tracking in cells and mice brains by intravital microscopy. Using NPs of different colors, we introduced a technique for barcoding of living cells. These NPs were also used as light-harvesting nanoantennas to prepare biosensors for cancer diagnostics and fluorescence in situ hybridization (FISH) for intracellular RNA imaging.
Photonic Instruments and Processes, ICube laboratory @ICube

R&D team: Photonic Instruments and Processes, ICube laboratory @ICube
Head: Sylvain Lecler
The IPP team (Photonic Instruments and Processes, ICube laboratory) has a long-standing expertise in microscopy and coherent imaging, as well as diffuse optics for surgical guidance. In particular, the team develops full-field OCT (scanning interference microscopy), endoscopic OCT, digital holography, hyperspectral imaging and quantitative phase imaging for biomedical application. Moreover, the team has a particular interest in multimodal imaging and contrast for example spectroscopy, elastography and Doppler imaging. The team has also more recently started work on fluorescence imaging, and in particular light-sheet microscopy for high-throughput imaging of live biomedical samples, based on the single-objective Oblique Plane Microscopy technique. The strategy of the team is to tackle biomedical questions with optical imaging with a multiscale approach from cellular scale to the organ scale.
Large complexes involved in transcription and translation team @IGBMC

R&D team: Large complexes involved in transcription and translation team @IGBMC
Head: Bruno Klaholz
As part of the Integrated Structural Biology platform at the Centre for Integrative Biology (hosting the national and European Infrastructures FRISBI, Instruct-ERIC and iNEXT-Discovery), and our research group at CBI/IGBMC we have recently done a series of developments in super-resolution fluorescence microscopy to facilitate single molecule localization microscopy (SMLM) analysis with the aim of integrating structural data generated through crystallography, cryo-EM, cryo-ET and FIB towards the cellular level (Biol Cell., 2017). We developed an integrated software for image reconstruction, drift & chromatic aberration correction, co-localization, resolution estimation (SharpViSu; Bioinformatics 2016), segmentation & clustering of labeled complexes [ClusterViSu;4], including 3D analysis and segmentation of SMLM data using Voronoi diagrams [3], as summarized in a book chapter [1]. Our latest development comprises a spectral demixing method which facilitates co-localization of proteins in SMLM (https://doi.org/10.1101/2021.12.23.473862).
Synthèse Pour l’Analyse (SynPA) team @IPHC

R&D team: Synthèse Pour l’Analyse (SynPA) team @IPHC
Head: Aline Nonat
The team is dedicated to the synthetic development of new tools for analytical and imaging applications. We notably develop luminescent labels for microscopy imaging and bio-analytical applications such as luminescence based assays. The team has a strong background in synthetic organic chemistry applied to the design of lanthanide based luminescent labels. Thanks to the spectral and temporal signature of lanthanide elements, the probes have exceptional characteristics such as very long lived excited states, line-like emission spectra and large pseudo Stokes’ shift. The main scientific axes related to the project are the design of luminescent probes displaying upconversion at the molecular and supramolecular level and the optimization of lanthanide based nanoparticles for time-resolved luminescence microscopy. Regarding upconversion, our molecular approach is expected to remove the troubles associated with the use of nanoparticles (biotoxicity, reproducibility,…) while profiting from the spectroscopic advantages of the anti-Stokes process. For lanthanide based nanoparticles, the displacement of the excitation wavelength towards the visible region will provide strong perspectives for luminescence microscopy and flow cytometry. Our team is fully equipped for organic and inorganic synthesis as well as for the spectroscopic characterization of the luminescent objects with UV-Vis-NIR absorption spectrometers and UV-Vis-NIR emission spectroscopy (up to 1600 nm) in the steady-state and time-resolved modes.
Key publications in 2022
|
|
|
|
|