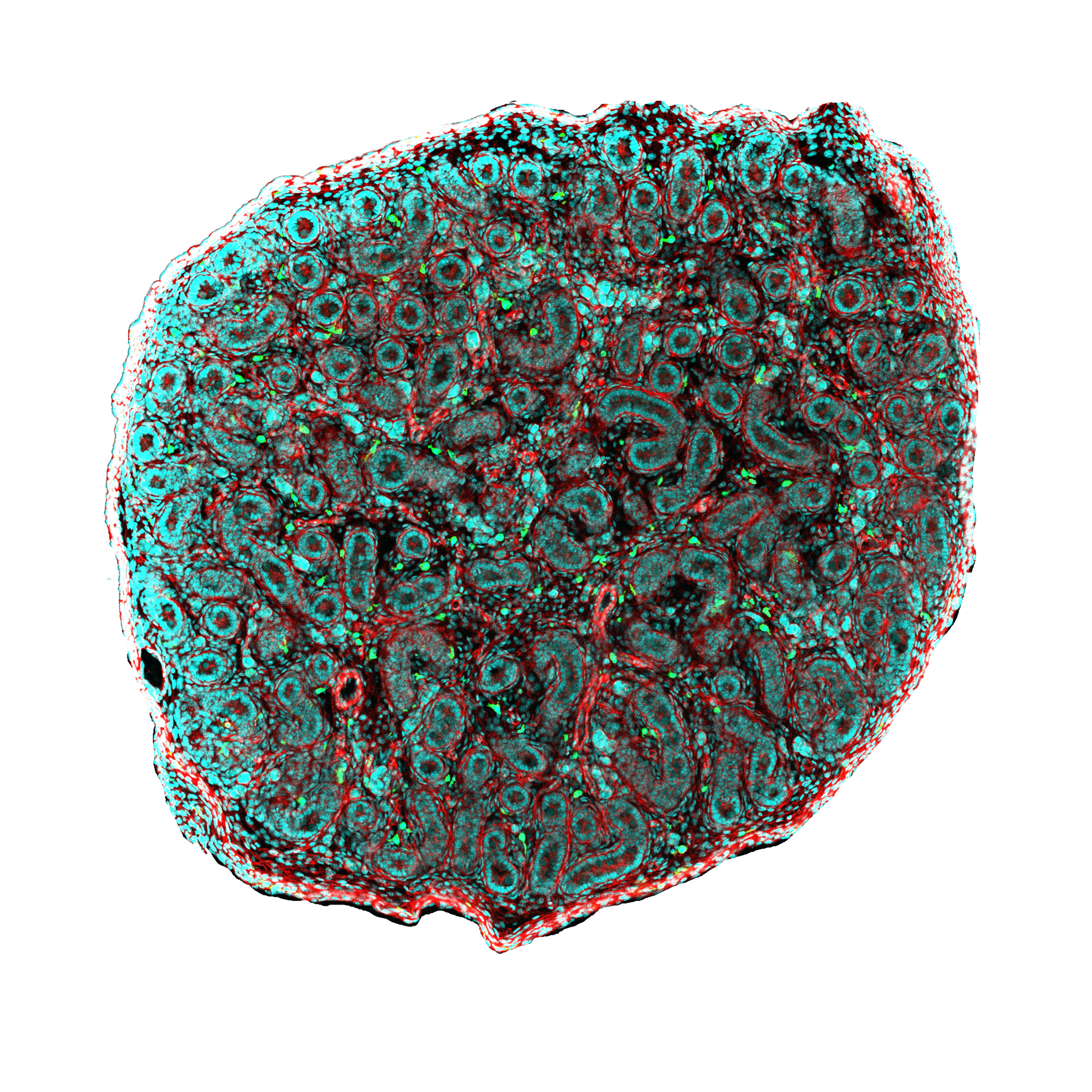
Imaging systems for dynamic and high resolution measurements in living organisms
The Marseille node gathers two institutes in life sciences (CIML and IBDM with a shared facility: PICsL) and one in photonics (Institut Fresnel), which aim at fostering new technologies and photonic innovation for cell imaging. Founded on strong scientific and technological collaborations and dynamic industrial partnerships, the Marseille node offers a variety of state-of-the art imaging systems (e.g. multiphoton, confocal imaging, light sheet microscopy, super-resolution microscopy, electron microscopy) to a large community of scientists in the field of cell developmental biology, immunology and neurobiology. The Marseille node has a strong expertise in designing and building cutting edge optical microscopy instruments.
Key expertise and services:
- single molecule detection and fluorescence fluctuation analysis,
- ultra-resolution microscopy and nanoscopy (PALM & STORM),
- light sheet microscopy, (iv) optical manipulation (nanodissection and optical tweezers),
- polarization resolved microscopy
- nonlinear multiphotonic microscopy including 2P and 3P fluorescence, harmonic generations and coherent Raman (CARS, SRS).
2021 in Numbers
- 172 hosted projects
- 45 publications
- 10 training programs
- 9 patents since 2011
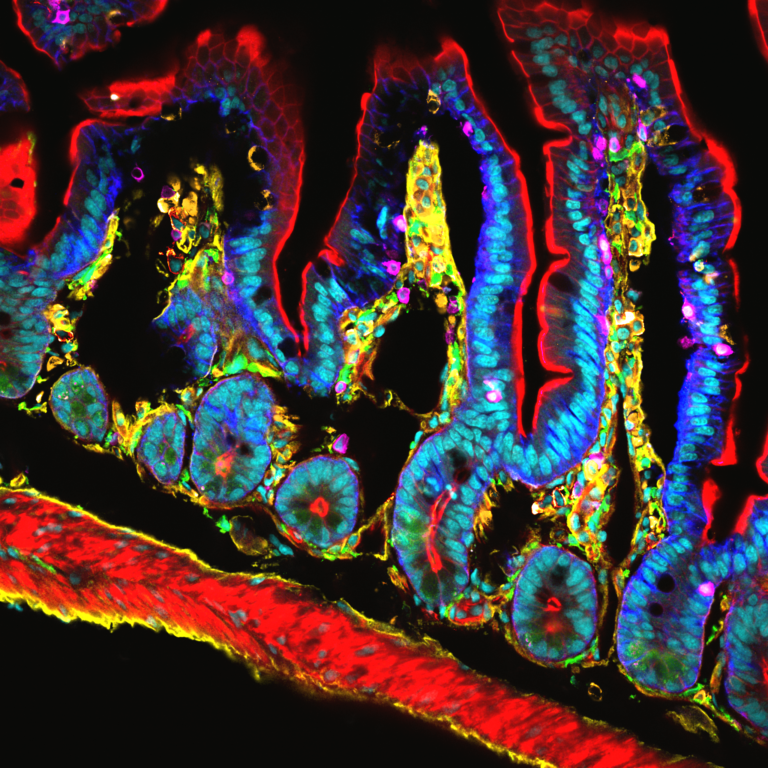
Technological Innovations
- Background free stimulated Rama spectroscopy (PRL, patent filled 2013)
- 2-Photon Lensless endoscope (Patents fille 2014-2016, Opt. Letters 2013)
- Laser manipulation of cells in vivo (PNAS 2015)
- Polarization resolved CARS (Nat. Comm., 2016)
- Polarization STORM (PANS 2016)
Tech transfer
- SRGold: Background free stimulated Raman (with APE GmbH)
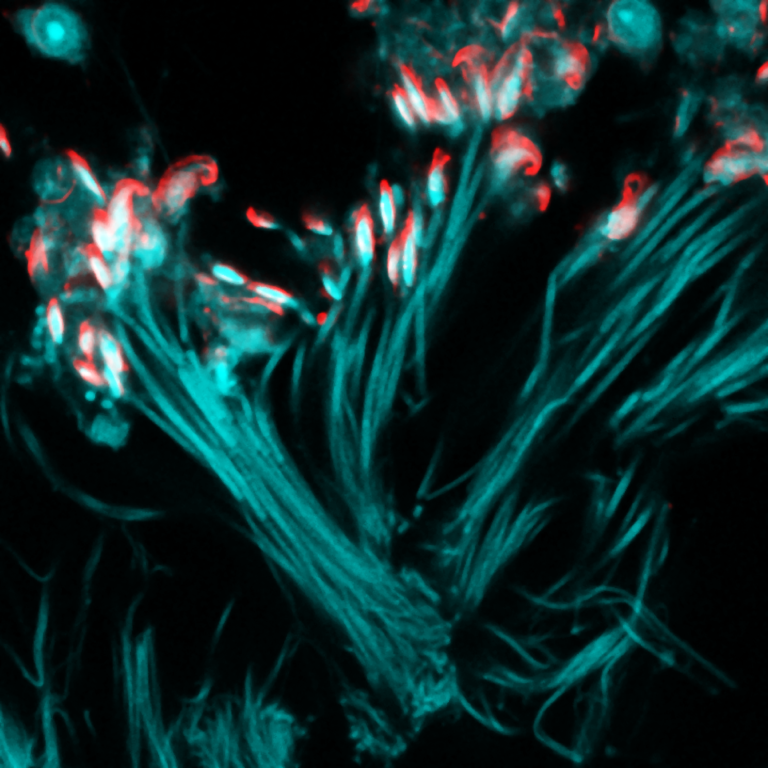
Most Innovative Systems Available for Booking
- Polar resolved spinning disk
- Spot variation FCS
- Two-photon wavelength mixing and non-linear contrats (CARS-SHG-THG)
- STED 3D
- Light sheet DSLM bessel beam
- Serial Block Face – Scanning Electron Microscope
PICsL (Shared Imaging Platform of Luminy Campus)

Facility: PICsL (Shared Imaging Platform of Luminy Campus)
Head: Pierre-François Lenne
The PICsL is located on the Luminy campus on two sites (IBDM and CIML ) that are within walking distance of each other. The PICSL offers a variety of state-of-the art imaging systems (multiphoton, confocal imaging, light sheet microscopy, super-resolution microscopy, electron microscopy). Mainly dedicated to developmental biology and immunology, the PICSL facility provides approaches for dynamic imaging, such as light sheet microscopy, super-resolution for tissue imaging (STED), multiphoton imaging but also more standard techniques such as time lapse imaging and confocal microscopy. Such methods are key to study quantitatively, the development of various animal species, and to decipher the operating mechanisms of the immune system, from molecules to whole organisms, such as Drosophila, C. elegans, Xenopus, chick, mice but also organotypic cultures and more recently organoids. The IBDM site also hosts an electron microscopy (EM) service. The EM service provides the scientific community with the expertise, material and electron microscopes necessary for molecular, cellular and tissue-level imaging by EM. Our service offer comprises the sample preparation with the following methods: negative staining, plastic embedding, immuno-labelling, high pressure freezing, freeze substitution, (cryo)-ultramicrotomy. We routinely use advanced imaging modes such as electron tomography (ET), Scanning Transmission Electron Microscopy (STEM), Scanning Electron Microscopy in « Serial Block-Face » mode, Correlative Light and Electron Microscopy (CLEM).
Microscopy systems available @PICsL
Services on this Facility
IMM Microscopy Core Facility
InMAGIC (INMED Imaging Center)

Facility: InMAGIC (INMED Imaging Center)
Head: François Michel
InMagiC (INMED iMAGing Center) is the microscopy platform of the Institute of Neurobiology of the Mediterranean: INMED in Marseille on the Luminy campus.
The platform is under the scientific direction of Rosa COSSART and under the responsibility of the engineer François MICHEL rosa.cossart@inserm.fr / francois.michel@inserm.fr
INMAGIC gathers in one place the multi-scale study tools necessary to study the development and the physio-pathological functioning of the central nervous system.
These tools cover the scales of observation from the whole animal (binocular loupes) to the dynamic morphology of neuronal contacts (synapses) thanks to widefield, confocal and multiphoton microscopes.
In addition, the platform provides a large number of training courses at all levels, both theoretical and practical, for all audiences (students, engineers, researchers, etc.).
The strong and original points of our structure are:
- A space dedicated to the techniques of tissue clearing and the acquisition of these samples by an ultramicroscope (light sheet) in platform access, as well as the expertise of the engineer in charge (active member of the GT transparencies of the RTMFM);
- The use of the multiphoton microscopy for the fast measurement (10-20 Hz) of the neuronal activity in vitro and in vivo on large populations of cells (up to more than 500 simultaneous cells).
Two high performance analysis stations are also available to all users. They include free and commercial software dedicated to analysis such as FIJI, ICY, IMARIS, Neurolucida360, Matlab…
Finally, the platform is a laureate of the PIA Equipex + 2020 and in this context the availability of tools beyond the state of the art in minimally invasive in vivo imaging will soon be open to the community.
These new tools will be commercial with industrial co-developments: a 3-photon excitation microscope, an ultra-fast AOD scanning microscope, a 2-photon endoscope or in academic development with the collaboration of the Fresnel Institute such as an ultra-thin endoscope and a deep recording station by photo-acoustics.
MOSAIC Group @Fresnel Institut

R&D team: MOSAIC Group @Fresnel Institut
Head: Hervé Rigneault
The Mosaic group of the Fresnel Institute headed by H. Rigneault has been involved for almost a decade in developing dedicated optical instruments for biological imaging. Among other, the team has developed together with CIML the “FCS diffusion law” approach in Fluorescence Correlation Spectroscopy that has been successfully applied to the cell membrane. More recently single particle tracking using multiple targets have proved to be powerful to distinguish confinement zone at the cell membrane and Holographic Optical Tweezers shows potential application into TCR/MHC control. Phase control for micro-manipulation and imaging is an active field of research at Mosaic. Since 2002, the Mosaic group has been involved in coherent Raman microscopy and nonlinear imaging and was the first in France to build and develop a CARS microscope. One of the group world recognized expertise is in polarization resolved fluorescence and nonlinear microscopy that has proved to be able to retrieve molecular order in cell and tissue imaging. The group is now also involved in the development of nonlinear imaging using endoscopes using innovative microstructured optical fibers. Another active field of research is fluorescence enhancement at the nanoscale using metallic nano-antenna that have the ability to perform dynamic analysis on time and spatial scales unreachable with far field optics.
Membrane dynamics and lymphocyte signaling Team @CIML

R&D team: Membrane dynamics and lymphocyte signaling Team @CIML
Head: Didier Marguet
Founded in 1976, the Centre d’Immunologie de Marseille Luminy (CIML) is a research institute internationally renowned in its discipline. From worm to man, from molecule to the whole organism, from physiology to pathology, the CIML addresses, over numerous models and scales, all fields of contemporary immunology: the genesis of different cell populations, their patterns of differentiation and activation, their implication in cancer, infectious and inflammatory diseases and the mechanisms of cell death. At CIML, Marguet team aims at understanding the role of membrane lateral dynamics and organization in T lymphocyte signaling, by analyzing the molecular interaction/association events at high spatial-temporal resolutions. A special emphasize is made at examination of the molecular dynamics in the plasma membrane to initiate and to integrate extracellular stimuli. In this context, Marguet team develops analytical methods based on the combination of single molecular sensitive detection approaches such as fluorescence correlation spectroscopy (FCS) and derivatives, of single particle tracking with optical tools allowing to manipulate the biological samples such as dynamic holographic optical tweezers.
Physical approaches to cell dynamics and tissue morphogenesis Team @IBDM

R&D team: Physical approaches to cell dynamics and tissue morphogenesis Team @IBDM
Team Leader: Pierre-François Lenne
The Developmental Biology Institute of Marseille (IBDM) is an international and interdisciplinary research institute oriented towards developmental biology and pathologies. The research activity is at the crossroads of development, neurobiology, cell biology, biophysics and genetics. The general theme of IBDM is to understand how the instructions encoded in the genome are translated to build structures (cells, tissues, organs) that perform specific functions, and how these processes are regulated and integrated in the whole organism. There are links between developmental biology and diseases such as cancer, neurodegenerative and genetic diseases. One of the priorities of IBDM is to foster interdisciplinarity through the integration of new and original approaches that create conceptual and technical interfaces. At IBDM, Lenne team aims at determining how (1) mechanical and (2) physical interactions are organized at cell surfaces in vivo and (3) how these interactions are processed to produce cell and tissue responses. To tackle these questions, we focus on two aspects of tissue morphogenesis, namely cell polarization and force transmission in fields of cells. We are using Drosophila and C. Elegans as systems to address questions (1-2) and question (3), respectively. The originality of our project relies in the integration of both physics and experimental biology to study quantitatively tissue morphogenesis.
Learning Meaningful Representations of Life
R&D team: Learning Meaningful Representations of Life
Team Leader: Paul Villoutreix
The team has close links with the Turing Center Multi-engineering platform where 3 engineers are specialized in image processing, database management and software development. The team develops inference
methods and machine learning tools for developmental biology, with the aim of understanding the relationships between morphogenesis and cell differentiation trajectories by bridging microscopy and single cell-omics.
Three expertise of the Team
- Image processing
- Machine learning
- Single cell omics
Key Publications
- V. Curcio, L. A. Aleman-Castaneda, T. G. Brown, S. Brasselet, M. A. Alonso, Birefringent Fourier filtering for single molecule Coordinate and Height super-resolution Imaging with Dithering and Orientation (CHIDO). arXiv:1907.05828 Nat. Communications 11 (1) (2020) https://doi.org/10.1038/s41467-020-19064-6
- Heuke, S. Sivankutty, C. Scotte, P. Stockton, R. A. Bartels, A. Sentenac, and H. Rigneault, “Spatial frequency modulated imaging in coherent anti-Stokes Raman microscopy,” Optica 7, 417-424 (2020). https://doi.org/10.1364/OPTICA.386526
- Clara Taffoni, Shizue Omi, Caroline Huber, Sebastien Mailfert, Mathieu Fallet, Jean-Francois Rupprecht, Jonathan J. Ewbank, and Nathalie Pujol, « Microtubule plus-end dynamics link wound repair to the innate immune response“, eLife, 9, (2020) DOI: 10.7554/eLife.45047
- Sebastien Mailfert, Karolina Wojtowicz, Sophie Brustlein, Ewa Blaszczak, Nicolas Bertaux, Marcin Łukaszewicz, Didier Marguet, Tomasz Trombik, “Spot Variation Fluorescence Correlation Spectroscopy for Analysis of Molecular Diffusion at the Plasma Membrane of Living Cells”, JoVE, 2020 http://doi.org/ 10.3791/61823
- Kaur S, Mélénec P, Murgan S, Bordet G, Recouvreux P, Lenne PF, Bertrand V. Wnt ligands regulate the asymmetric divisions of neuronal progenitors in C. elegans embryos. Development. 2020 Apr 6;147(7):dev183186. DOI: 10.1242/dev.183186 PMID: 32156756