A custom-made High-Content Screening method to identify therapeutic drugs for a fatal neurodegenerative disorder
High-content screening (HCS) is a technology used in drug discovery and research to analyze cell phenotype. HCS has created new opportunities for studying biological phenomena as it combines high-throughput screening methods with automated microscopy on microplate format. Using automated cell manipulations and microscopy platforms, it is possible to easily screen the effects of selected drugs on the model’s phenotype. This technology, available at France-BioImaging at Montpellier Ressources Imaging (MRI), has been used in a recent study identifying therapeutic drugs for a fatal neurodegenerative disorder, the Giant Axonal Neuropathy (GAN). Benoît Bordignon and Cédric Hassen-Khodja, screening experts at the MRI and co-authors in this recent publication, tell you more about the reasons why the HCS technology was essential!
Get routinely quick acquisition of numerous samples with HCS
As mentioned before, High-Content Screening uses automated cell manipulations and automated microscopy platforms to easily visualize the modification of the phenotype. In other words, it allows image acquisitions and analysis of a large number of samples, and collects data of biological parameters of interest directly for statistical analysis. Image acquisition and analysis are done in batches and rapidly, which limit potential bias as the process will be identical from one well to another. A huge advantage that prevents issues and saves time!
Imaging can help therapeutic research
Giant axonal neuropathy (GAN) is a fatal neurodegenerative disorder for which there is currently no treatment. Affecting the nervous system, GAN starts in infancy with motor deficits that rapidly evolve toward total loss of ambulation. Using the gan zebrafish model that reproduces the loss of motility as seen in patients, scientists conducted the first pharmacological screening for the GAN pathology. They combined behavioral, in silico, and high-content imaging analyses to identify drugs restoring locomotion, axonal outgrowth, and stabilizing neuromuscular junctions in the gan zebrafish.
This is when imaging becomes essential! To validate at cellular level the last 103 selected hits, they have developed a custom-made High-Content Screening method to check if the phenotype was also restored at the cellular level in gan zebrafish, in particular on neuromuscular junction (NMJ) and axonal function. The postsynaptic nature of the drug’s cellular targets provides direct evidence for the pivotal role the neuromuscular junction holds in the restoration of motility.
The results of this study identify the first drug candidates that can now be integrated in a repositioning approach to fasten therapy for the GAN disease.
Scientists working on this study therefore created an automated image analysis protocol using HCS to quantify neuromuscular junctions’ number/size and measure axon length in treated gan zebrafish. Thus, image data treatment had a central place in this study. And this is just an example! This novel high-content imaging methodology represents a useful automated procedure that can be transferred to other neuromuscular conditions for mechanistic studies and drug screening.
Thanks to Benoît Bordignon and Cédric Hassen-Khodja for providing very helpful insights of the study!
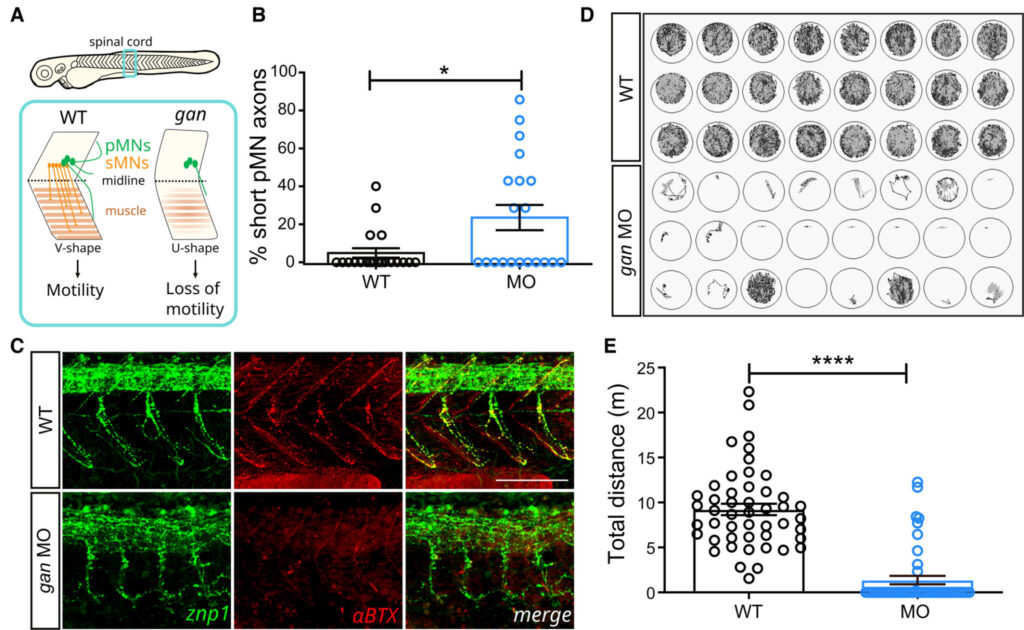
A – Schematic of the behavioral and cellular defects described in the gan zebrafish model. Underlying the loss of motility in the gan zebrafish, the architecture of the spinal cord is remodeled with shortening and/or absence of MN axons (in green, primary MNs; in red, secondary MNs) and loss of neuromuscular junctions (NMJ). The neuromuscular phenotype is accompanied by a change in the shape of myofibers, which adapt a “U-shape” instead of a normal “V-shape.”
B – The percentage of shorter pMN axons (< 70 μm) per fish is significantly higher in gan morphants (n = 20) than in noninjected WT (n = 20) at 48 hpf.
C – Representative images for the neuromuscular junctions (znp1: green; αBTX: α-bungarotoxin: red) in WT and gan morphants at 48 hpf. Note the shorter pMN axons and sparse AChR clusters in gan morphants.
D – Representation of the cumulative tracking of the spontaneous locomotion of 5-day-old larvae for 1 h, in noninjected and MO-injected animals.
E – Quantitative measures of the traveled distance (m: meter) show total loss of locomotion in 79.2% of gan morphant; n = 48 (WT), n = 48 (MO).
Get access to one of our services!
You need HCS or another imaging technology or expertise that France-BioImaging provides? To get open access, please login via Euro-BioImaging website! You just have to choose the technology you want to use, then submit your proposal. All applications will be processed by the Euro-BioImaging Hub in close relation with France-BioImaging. And of course, all scientists regardless of their affiliation, area of expertise or field of activity can benefit from open access services! Users whose projects will be validated by Euro-BioImaging will benefit from a waiver for the access cost on France-BioImaging core facilities (france-bioimaging.org/access)
Lescouzères, L., Hassen‐Khodja, C., Baudot, A., Bordignon, B., & Bomont, P. (2023). A multilevel screening pipeline in zebrafish identifies therapeutic drugs for GAN. EMBO Molecular Medicine, e16267. https://doi.org/10.15252/emmm.202216267