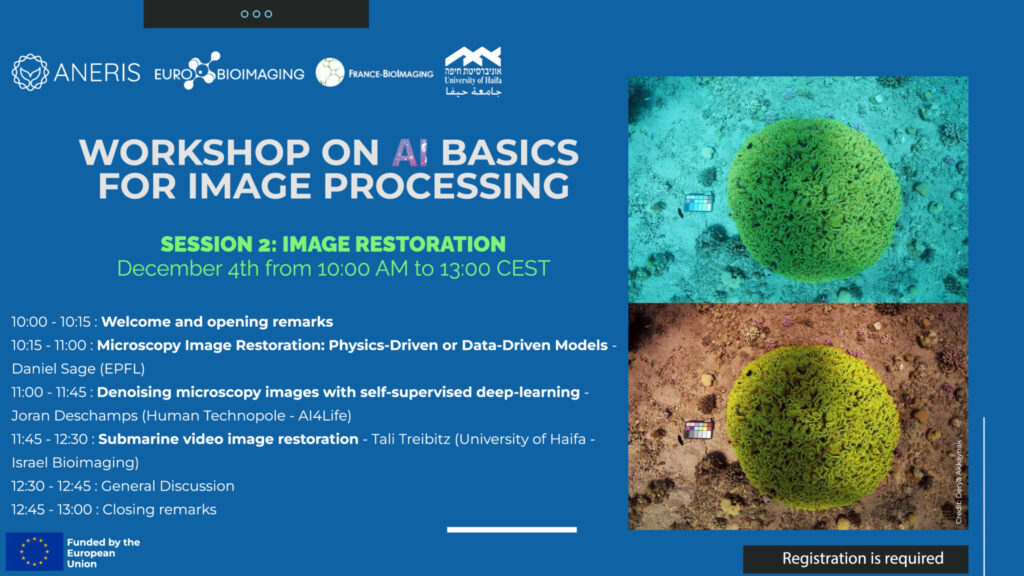
This school aims at training students and researchers to master fluorescent markers used in advanced fluorescence bioimaging: their diversity, how they actually work and what are their current development.
It will be organized at the Ecole de Physique des Houches, in the french Alps, from March 17th to 22th, 2024.
Objectives of the school :
Fluorescence microscopy techniques play an essential role in biology and medicine. A very rapidly developing field concerns nanoscale imaging (“nanoscopy”), which today ties together different fields of biology such as structural and cell biology. Similarly, “single molecule” imaging and spectroscopy techniques are in rapid development and play a central role in life science research. All fluorescence microscopy methods are based on labelling samples with appropriate markers: fluorescent proteins, organic fluorophores or nanoparticles. All these type of markers are characterized by very complex photophysical behaviors, leading to blinking, chromatic conversion, photoreactivity, etc… However, nanoscopy and single molecule spectroscopy or imaging, specifically rely on the ability to understand and control these properties.
The school we offer is motivated by the fact that more and more laboratories around the world are committed to implementing advanced microscopy techniques, especially super-resolution, with an increasing number of dedicated platforms. However, there is a lack of overall knowledge about the selection of fluorescent markers, their properties and mechanisms, and the type of artifacts they can create. The school’s objective, through interdisciplinary teaching between physics, chemistry and biology, is to overcome this gap by transferring knowledge to the participants’ laboratories. Our school also aims to make participants aware of new developments in the field in order to prepare them to become major players in the breakthroughs that may result from them.
Invited Teachers
Confirmed teachers for the 2024 edition
Dominique Bourgeois, Institut de Biologie Structurale, CNRS, Grenoble, France
Claire Deo, EMBL Heidelberg, Germany
Ulrike Endesfelder, Bonn University, Germany
Viktorija Glembockyte, LMU Munchen Germany
Mike Heilemann, Goethe Universitat Frankfurt, Germany
Ludovic Jullien, Sorbonne University and ENS Paris, France
Doory Kim, Hanyang University, Korea
Andrey Klymchenko, CNRS, Université de Strasbourg, France
Emmanuel Margeat, Centre de Biologie Structurale, CNRS, Montpellier, France
Ivana Nikic-Spiegel, U. Tuebingen, Germany
Franck Riquet, Lille University, France
Markus Sauer, U. Würzburg, Germany
Nathan Shaner, UCSD, USA
Alison Tebo, HHMI Janelia Research campus, USA
Organizers
Dominique Bourgeois, Institut de Biologie Structurale, Grenoble, France
Claire Deo, EMBL Heidelberg, Germany
Ulrike Endesfelder, Bonn University, Germany
Emmanuel Margeat, Centre de Biologie Structurale, Montpellier, France
More information and registration on: https://fluorescenceleshouches.wordpress.com/