News from FBI-Montpellier node
The R&D division of FBI-Montpellier is focused on the development of super-resolution and fluctuation microscopy methods. On the super-resolution front, we have recently developed a new instrument for the rapid acquisition of single-molecule localization microscopy (SMLM) images of thick intracellular structures (>5µ) at nanometer resolutions without scanning.
In conventional SMLM, the gain in resolution arises from the precise localization of single emitters labeling the structure of interest, thus enabling the reconstruction of images in 2D with a resolution of ~10-20 nm. Most of the biological structures are, however, three-dimensional. To increase the axial depth while conserving spatial resolution, we combined two ingredients. First, we used multi-focus microscopy (MFM) (Abrahamsson, 2013), a technology that allows for the simultaneous acquisition of several image planes on the same camera chip. We combined MFM with point-spread function (PSF) engineering, a method that relies on the use of asymmetric PSFs to enable axial localization. For this development, we designed and built binary multifocus gratings with ~ 400 nm spacing, ideal for SMLM intracellular imaging of eukaryotic cells using organic dyes or photo-activatable proteins. Our method requires only the detection and localization of emitters in a single imaging plane, thus allowing for an increase in the distance between MFM planes to reach thicker axial imaging depths. Importantly, our method also allows for a considerable increase in image reconstruction speed without sacrificing localization precision, as it requires the fitting of the emitter PSF in a single plane to yield a 3D localization. This development led to a Patent application filing (European Patent EP15305787.2 filed on May 26, 2015) and a publication (Oudjedi, 2016) (Figure 1).
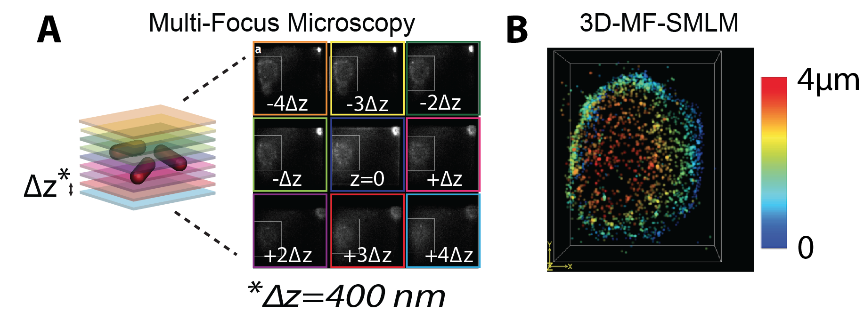 Figure 1: (A) Multi-focus microscopy (MFM) allows for the instantaneous acquisition of whole nuclei in a single camera frame. (B) Reconstruction of the nuclear envelope of a S2 Drosophila cell with >4µm depth of field at nanometer resolutions can be achieved with our microscope, 10-100 times faster than conventional 3D-SMLM.
Figure 1: (A) Multi-focus microscopy (MFM) allows for the instantaneous acquisition of whole nuclei in a single camera frame. (B) Reconstruction of the nuclear envelope of a S2 Drosophila cell with >4µm depth of field at nanometer resolutions can be achieved with our microscope, 10-100 times faster than conventional 3D-SMLM.
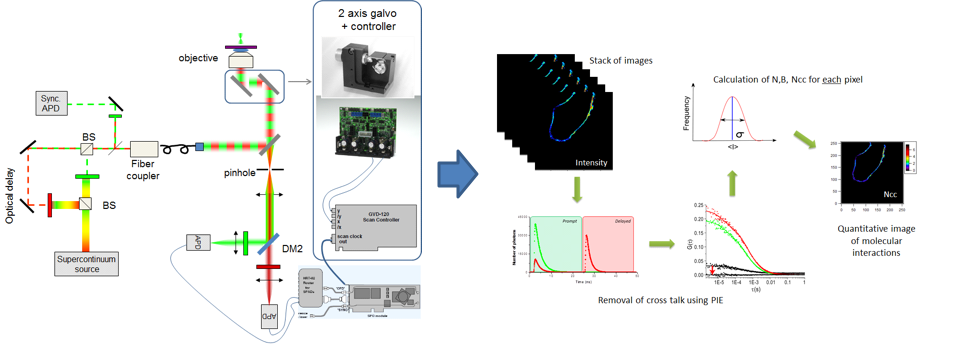
On the fluctuation microscopy front, we have developed a method to measure protein concentration, diffusion coefficient and brightness for low photon flux fluorophores and eliminating cross-talk between channels. Fluorescence correlation spectroscopy (FCS) techniques allow for the determination of the concentration (N), the diffusion coefficient (D), and the brightness (B) of fluorescent molecules of interest, and thus report on their oligomerization properties and interactions with cellular components (Figure 2). However, FCS measurements are traditionally disturbed by a low photon flux (especially under two-photon excitation), strong photobleaching, and cross-talk between spectrally distinct detection channels. Recently, thanks to a CNRS “Instrumentation aux limites” funding, we have developed a homemade microscope that will overcome all these limitations (Hendrix, 2014), by combining : (1) A pulsed supercontinuum source allowing great versatility of choice of colors of excitation, and therefore of fluorophores used, and an increase in the photon flux, thus improving the signal / noise ratio; (2) An alternating laser excitation scheme (Olofsson, 2013) coupled to a dual-channel TCSPC detection card, to eliminate cross-talk effects; and (3) A laser scanning galvanometric system to reduce photobleaching, and obtained spatially resolved Number, Brightness, and Cross-interaction maps in living cells.
References
Abrahamsson, S., Chen, J., Hajj, B., Stallinga, S., Katsov, A. Y., Wisniewski, J., … Gustafsson, M. G. L. (2013). Fast multicolor 3D imaging using aberration-corrected multifocus microscopy. Nature Methods, 10(1), 60–63.
Hendrix J., Lamb D.C. Implementation and Application of Pulsed Interleaved Excitation for Dual-Color FCS and RICS (2014). In Fluorescence Spectroscopy and Microscopy: Methods and Protocols, Methods in Molecular Biology. 1076, 371-417
Olofsson L., Margeat E. Pulsed interleaved excitation fluorescence spectroscopy with a supercontinuum source (2013). Optics Express, 21(3), 3370-8
Oudjedi, L., Fiche, J.-B., Abrahamsson, S., Mazenq, L., Lecestre, A., Calmon, P.-F., … Nöllmann, M. (2016). Astigmatic multifocus microscopy enables deep 3D super-resolved imaging. Biomedical Optics Express, 7(6), 2163.