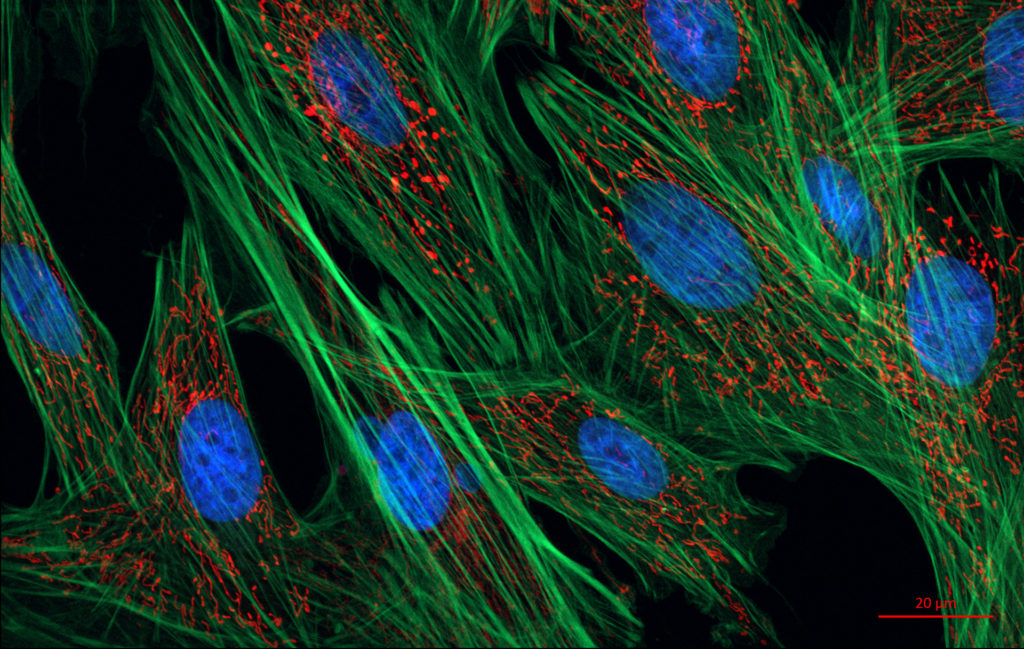
Cutting-edges methods in image-based phenotyping and multi-scale tracking of therapeutic agents
The Bretagne-Loire Node is composed of three cellular and tissue imaging platforms: H2P2, MicroPICell and MRic-of the Bioimaging axis of GIS Biogenouest and research and development teams (belonging to IGDR, Numecan, Cesam, Inria-Rennes, LS2N, SFR F. Bonamy) that support the activities of these platforms.
Located in Nantes and Rennes, the three platforms have the IBiSA certification and their expertise in life imaging and pathological anatomy are complementary.
The mission of the node is to provide the latest technological and methodological advances in microscopy for an integrated understanding of cellular and tissue activities in particular for preclinical research.
The Bretagne-Loire Node has expertise in technology transfer and translational research and benefits from a privileged scientific environment due to its affiliation with the University Hospital Centres.
2021 in Numbers
- 589 hosted projects
- 19 publications
- 12 training programs
- 8 patents since 2011
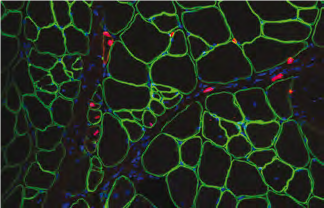
Technological Innovations
Correlative microscopy (Nature Methods 2017, Burel 2018 )
Multimodal Fluorescent organic nanoassemblies (Small 2018)
High-content screening Fluorescence Life Time, High content screening with large field of views
Label free multimodal imaging (Raman , Infra red, CARS, SHG)
Multiplexing staining
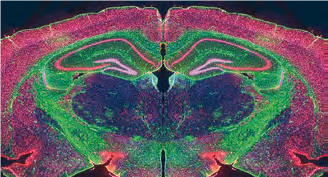
Tech transfer
Roboscopy (Inscoper, Photonlines)
Episcopic microscopy for 3D histology (Kaerlabs)

Most Innovative Systems Available for Booking
Light-sheet microscopy
TEM and cryo-microscopy
Raman microspectroscopy
Micropatterning
Structure illumination microscopy, STORM
H2P2 (Histo pathology High precision)

Facility: H2P2 (Histo pathology High precision)
Head & CoHead: Anthony Sébillot, Bruno Turlin and Marie-Dominique Galibert
The H2P2 platform performs histology work on human, animal and plant tissues. The facility is equipped with last generation devices to increase analysis reproducibility and ensure rapid automatic processing. We produce Tissues Micro Array (TMA) that can group hundreds of tissulars spots on a single microscope slide. We have developed a thorough expertise in immuno-labeling (more than 1000 Ac in our catalog) as well as multiplex staining, a technique that can highlight up to 6 different proteins per slide, fluorescence or chromogenic. Since 2021, with the CELLDive technology we are able to visualize up to 60 markers on the same sample. To visualize slides we offer several options: epifluorescence microscope, virtual slides from a Hamamatsu scanner with fluorescence and a new generation confocal scanner (3D-Histech) with seven channels in fluorescence. This latter technology gives the opportunity to scan thick tissues (300µm) after clearing. Slides are analysed in 2D using Halo, an image analysis platform/software (with machine learning) for quantitative tissue analysis in digital pathology. We perform 3D images routinely and advanced analysis reconstruction with Amira. We also developed extensive expertise in laser capture microdissection, allowing single cells capture. We are developing in situ molecular technologies, as Raman micro spectroscopy imaging on biological samples with statistical analysis of obtained spectra.
Services on this Facility
MicroPICell (Tissue and cell imaging)

Facility: MicroPICell (Tissue and cell imaging)
Head: Perrine Paul-Gilloteaux, Vincent Guen
MicroPICell is an imaging facility specialized in sample preparation (histology), cellular and tissular imaging (photonic microscopy) and data processing and analysis (bioimage analysis). The facility is located within the precinct of the Hotel Dieu hospital, Nantes University. The platform is affiliated to the Structure Federative de Recherche UMS INSERM 016/CNRS 3556/Nantes University and teamed up with Nikon Instrument society and the APEX veterinary facility to create the Center of Excellence Nikon Nantes in 2016. The MicroPICell facility offers a complete integrated cellular imaging service to all users after analysis of the feasibility of their projects. The applications range is then very large, for example cancer research and immunology, regenerative medicine or stem cells research. It is organized in 3 main services: histology, photonic microscopy, image data management and analysis, all these services working closely together on users projects.
MRIC (Microscopy Rennes Imaging Center)

Facility: MRIC (Microscopy Rennes Imaging Center)
Head: Marc Tramier, Claude Prigent
The platform provides regulated access to imaging systems, as well as scientific and technical assistance in the design of projects and experiments. Proposed techniques are: epifluorescence, video microscopy, confocal, spinning disk, multiphoton, lightsheet, TEM, cryo-electron microscopy, high pressure freezing, tomography, immuno-electron microscopy.
Backed by research teams, the platform also has a strong R&D activity dedicated to the design of prototype microscopes. Finally, MRic provides training to microscopy techniques.
CeDRE (Cell Division Reverse Engineering)

R&D team: CeDRE (Cell Division Reverse Engineering)
Head: Jacques Pécréaux
MFQ (Microscopie de Fluorescence Quantitative)

R&D team: MFQ (Microscopie de Fluorescence Quantitative)
Head: Marc Tramier
The team aims at developing techniques and methodologies in fluorescence microscopy to study dynamics of protein-protein interactions and biochemical activities in live sample. Team approaches are mainly driven by methodological and technological development, its transfer and its applicability in biology to answer relevant new questions. Recently, the team is investigating the possibility to implement smart and autonomous microscopy approches to be able to develop robust and automated high content microscopy pipelines of cutting-edge methods such as F-microscopy or photoperturbative experiments.
Multifunctional molecular and hybrid nanomaterials @CEISAM
R&D team: Multifunctional molecular and hybrid nanomaterials @CEISAM
Head: Eléna ISHOW
Nanomaterials nowadays occupy an indisputable place for their diagnostic and therapeutic potentialities, offering new paradigms in the field of medicine in terms of follow-up, safety and personalized treatments. Due to the complexity of the biological environment, mastering the synthesis and understanding the interplay between nanomaterials and biological entities, especially cells, is an imperative need to overcome numerous pitfalls related to toxicity, chemical instability, undesirable targeting, diagnostic artefacts and bioaccumulation, to cite only the major ones.
To this aim, we have been developing for several years multimodal nanoassemblies, originally based on small-molecule photoactive materials, self-assembled as a platform and comprising functional inorganic nanoparticles to bridge the gap from in cellulo to in vivo investigations. Their versatile fabrication offers straightforward architectural tunability, allowing us to decipher their in cellulo interactions.